🧵 CESR Path Series Ep. 3: How to Gather Evidence of Research Experience
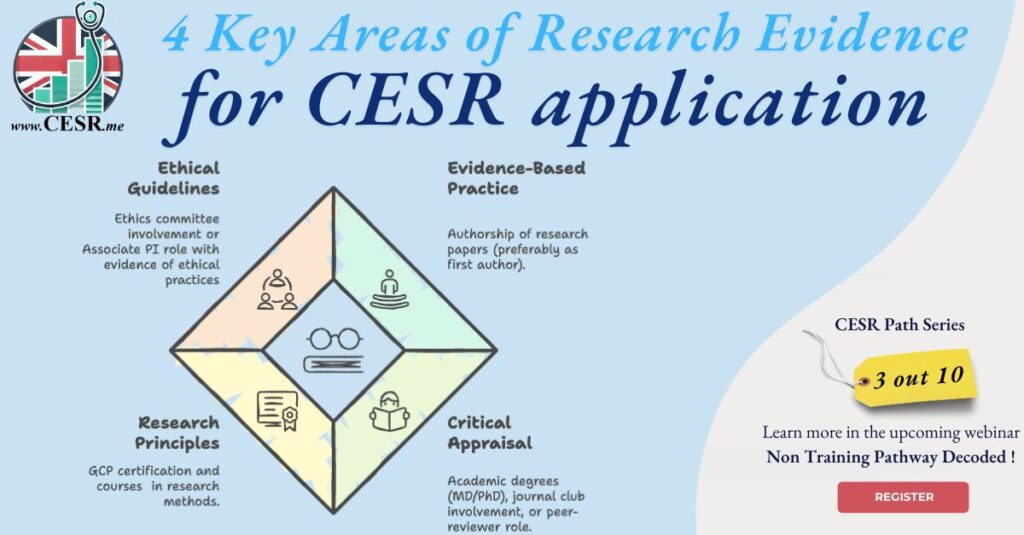
When pursuing a Certificate of Eligibility for Specialist Registration (CESR), one of the key areas to address is your research experience. Building a comprehensive portfolio in this domain can enhance your application significantly. While none of the following elements are mandatory, including as many as possible strengthens your case. Here’s how you can gather and present evidence of research experience effectively.
1. Demonstrating Evidence-Based Practice
Active engagement in research showcases your ability to contribute to the medical field. Here’s how you can demonstrate this:
- Authorship of Papers: Aim to be a substantive contributor, ideally as the first author, on published papers.
- Presentations: Provide proof of poster or podium (oral) presentations at national or international meetings where you were the presenter.Tip: Keep a record of conference abstracts and presentation certificates. These documents add substantial weight to your application.
2. Understanding Critical Appraisal of Literature
Your ability to critically evaluate and synthesize research is another important competency to demonstrate:
- Academic Degrees: Holding an MD or PhD provides strong evidence of critical appraisal skills.
- Journal Club Participation: Document your involvement in regular journal club discussions.
- Reviewer Role: Submit evidence of serving as a reviewer for medical journals, particularly in your specialty, such as neurosurgical journals.Why This Matters: These activities reflect critical thinking and a thorough understanding of current medical literature.
3. Applying Basic Research Principles
Showcase your foundational knowledge of research methodologies through:
- Good Clinical Practice (GCP) Certification: This certification demonstrates your understanding of clinical research standards and ethical practices.
- Research Methodology Courses: Include certificates or evidence of training in research design and analysis.
4. Understanding Research Governance and Ethical Guidelines
Demonstrate your knowledge of conducting research responsibly and ethically:
- Ethics Committee Involvement: Provide evidence of participation in an ethics committee or applications for ethical approval of your research.
- Associate Principal Investigator (PI) Role: Document your involvement as an Associate PI in clinical trials, including evidence of managing informed consent procedures.
Pro Tip: Organize your research evidence systematically. Use certificates, letters of involvement, and published work to clearly demonstrate your contributions.
The Journey of 10 steps :
1. 🚀 CESR Main Considerations: Starting the Journey
2. How to Gather Evidence of Teaching Experience
3.🧵 CESR Path Series : How to Gather Evidence of Research Experience
4.🕒 CESR Application Timeline: Key Stages and Expectations
5. 🏛️ How to Choose Your Referees for Your CESR/Portfolio Application
6.Ward-Based Assessment (WBA): A Comprehensive Guide
7. Writing Effective Reflection in CESR Applications
8. Mastering Your Logbook in CESR application
9.How to Provide Quality Improvement Evidence for CESR Application
10.Building a Strong Portfolio of Management and Leadership Evidence for CESR Application
